Abstract
Background: Proteasome inhibition (PI) is the backbone of various anti Multiple Myeloma (MM) treatment regimens. Initially, patients are well responsive, however, over time virtually all patients develop PI resistance. The upregulation of the multidrug resistance protein (MDR1/ABCB1) has previously been demonstrated to induce Carfilzomib (CFZ) resistance in cell lines and patients (Besse et al., Leukemia, 2018). However, the underlying molecular mechanisms are not yet fully understood. In this work, we investigated, whether DNA methylation may underlie the upregulation of ABCB1 in vitro and in vivo.
Methods: First, we confirmed the regulatory impact of DNA methylation in the targeted ABCB1 promoter region using a CpG-free vector-based dual luciferase reporter assay. The methylation level of the same region was then assessed via Deep Bisulfite Sequencing (DBS) in the four PI-sensitive MM cell lines ARH77, L363, AMO1, and RPMI8226 along with their respective CFZ resistant derivatives. In addition, we screened 15 newly diagnosed MM (NDMM), 50 relapsed/refractory MM (RRMM), and 25 plasma cell leukemia (PCL) patients. Finally, the Proteomes of MM cell lines were measured by micro-LC-Orbitrp MS/MS and searched against human Swiss-Prot database by MaxQuant.
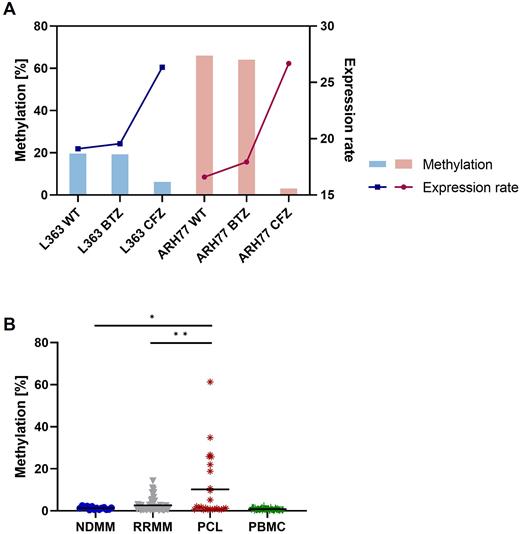
Results: In cell lines AMO1 and SH-SY5Y, hypomethylation of the target region increased the luciferase activity by 27- and 130-fold, indicating that the methylation of the investigated ABCB1 promoter region downregulates the gene expression. In addition, two out of four CFZ-resistant derivatives showed a dramatically decreased ABCB1 promoter methylation rate compared to their parental CFZ-sensitive cells (methylation degree: ARH77 WT 66% / ARH77 CFZ 2.9%, L363 WT 19.6% / L363 CFZ 6.2%). Of note, our proteomic screen revealed the ABCB1 expression level of our CFZ-resistant ARH77 and L363 models to be significantly increased, escalating from 16.5 to 26.6 (log2 fold) in ARH77 and 19.1 to 26.3 (log2 fold) in L363, demonstrating a negative correlation with the obtained promoter methylation level (Figure A).
In patients, the ABCB1 promoter methylation rate increased from NDMM (mean: 1.32% ± 0.67%, min: 0.5%, max: 2.5%) to RRMM patients (mean: 2.66% ± 3.21%, min: 0.3%, max: 14.3%). PCL patients (mean: 10.24 % ± 15.10%, min: 0.4%, max: 60.9%) showed significantly higher methylation levels compared to RRMM (p ≤ 0.01). Lowest values were assessed in 47 paired PBMCs from NDMM and RRMM (mean: 0.82% ± 0.48%, min: 0.2%, max: 2.5%). Notably, 10 out of 50 RRMM patients were CFZ-resistant, however, their ABCB1 methylation level did not differ significantly from the levels obtained from CFZ-sensitive patients (mean: 2.68% ± 2.79%, min: 0.4%, max: 8.2%) (Figure B). Of note, there were no significant differences between primary and secondary PCLs (p = .8151) but a tendency for decreased methylation in samples derived from peripheral blood compared to samples obtained from the bone marrow (p = .0728).
Conclusions: Based on our dual luciferase assay and DBS, we are first to confirm regulatory effects of ABCB1 promoter hypomethylation on the expression of ABCB1 in two MM cell line models, which may underlie their CFZ resistance. On the contrary, in patients, we observed an increasing methylation rate of the ABCB1 promoter region from NDMM to RRMM and a significantly increased methylation in PCL. However, no difference was seen between CFZ-resistant and sensitive patients.
Disclosures
Kull:Takeda: Honoraria, Other: Sponsored travel; Oncopeptides: Honoraria; Celgene: Other: Sponsored travel; Janssen: Other: Sponsored Travel. Raab:Amgen: Membership on an entity's Board of Directors or advisory committees; BMS: Membership on an entity's Board of Directors or advisory committees; Heidelberg Pharma: Research Funding; Novartis: Membership on an entity's Board of Directors or advisory committees; Sanofi: Membership on an entity's Board of Directors or advisory committees; Takeda: Membership on an entity's Board of Directors or advisory committees. Einsele:BMS/Celgene: Consultancy, Membership on an entity's Board of Directors or advisory committees, Other: Travel Grants, Research Funding; Janssen: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Other: Travel Grants, Research Funding; Amgen: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Other: travel grants; Takeda: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Other: travel grants; Sanofi: Consultancy, Honoraria, Research Funding; GSK: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; Novartis: Other: travel grants. Rasche:Pfizer: Honoraria, Membership on an entity's Board of Directors or advisory committees; Sanofi: Honoraria, Membership on an entity's Board of Directors or advisory committees; GSK: Honoraria; Amgen: Honoraria, Membership on an entity's Board of Directors or advisory committees; BMS: Honoraria, Membership on an entity's Board of Directors or advisory committees; Janssen: Honoraria, Membership on an entity's Board of Directors or advisory committees. Kortuem:Janssen: Research Funding; Janssen, BMS, GSK, Abbvie, Pfizer: Consultancy.
Author notes
Asterisk with author names denotes non-ASH members.